|
Ig-seq applied to human B cells has potential in a variety of applications ( 4), particularly in antibody drug discovery ( 5– 7), profiling for vaccine development ( 8, 9), and biomarker-based diagnostics ( 10, 11). Due to rapid technological advances, high-throughput sequencing of Ig genes (Ig-seq) has become a major approach to cataloguing the diversity of antibody repertoires ( 1– 3). In-depth and accurate characterization of these repertoires provides valuable insight into the generation and maintenance of immunocompetence, which can be used to monitor changes in immune status, and to identify potentially reactive clones for therapeutic or other uses. Unique V(D)J recombination events in differentiating B cells create a highly complex repertoire of receptors, which is shaped upon antigen experience to produce the more targeted, high-affinity memory BCR network. In particular, B cell antigen receptors (BCRs, and their analogous secreted form, antibodies) are generated upon rearrangement of incomplete, germline-encoded variable (V), diversity (D, heavy-chain only), and joining (J) gene segments. A hallmark of these cells is the somatic generation of clonally unique antigen receptors during primary lymphocyte differentiation. We anticipate that our standards and error and bias correction pipeline will become a valuable tool for researchers to validate and improve accuracy in human Ig-seq studies, thus leading to potentially new insights and applications in human antibody repertoire profiling.Īdaptive immune responses are governed by cooperative interactions between B and T lymphocytes upon antigen recognition. This strategy enabled robust and reliable estimation of key repertoire features such as clonotype diversity, germline segment, and isotype subclass usage, and somatic hypermutation. Finally, we performed Ig-seq on purified human circulating B cell subsets (naïve and memory), combined with a cellular replicate sampling strategy. Next, we adapted a library preparation protocol that incorporates unique molecular identifiers (UIDs) for error and bias correction which, when applied to the synthetic standards, resulted in highly accurate data. First, we designed a set of 85 synthetic antibody heavy-chain standards ( in vitro transcribed RNA) to assess correction workflow fidelity. Here, we have established an approach for performing highly accurate human Ig-seq by combining synthetic standards with a comprehensive error and bias correction pipeline. However, a major challenge in Ig-seq is ensuring accuracy, as library preparation protocols and sequencing platforms can introduce substantial errors and bias that compromise immunological interpretation. When applied to human repertoires, Ig-seq provides insight into fundamental immunological questions, and can be implemented in diagnostic and drug discovery projects.  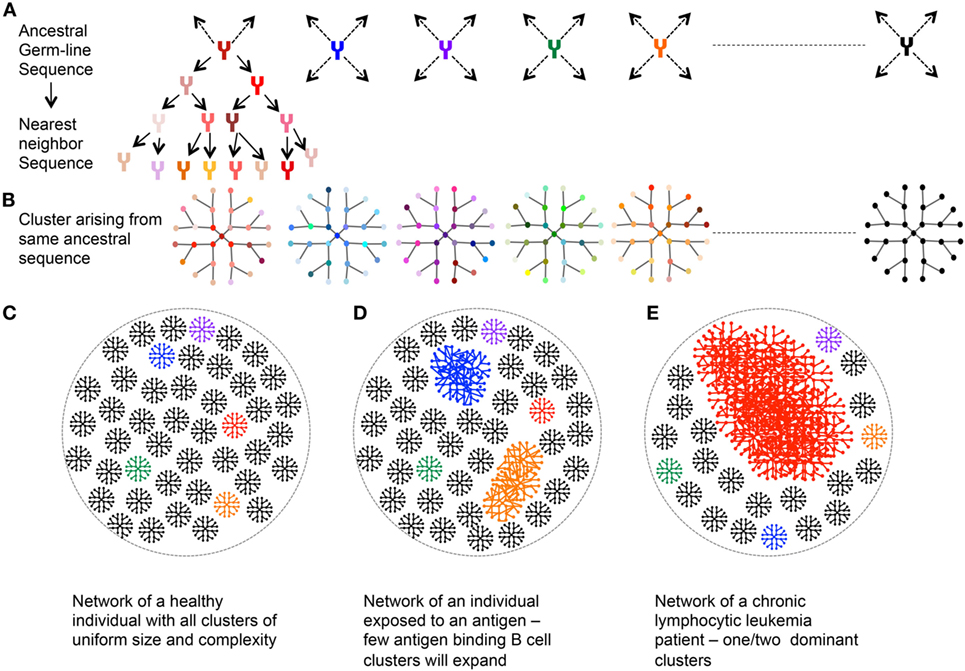 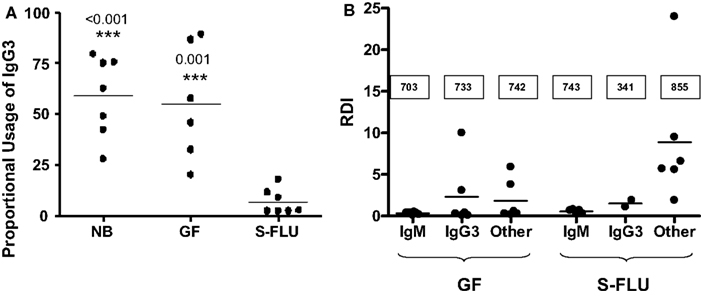
High-throughput sequencing of immunoglobulin (Ig) repertoires (Ig-seq) is a powerful method for quantitatively interrogating B cell receptor sequence diversity. 2Novartis Institutes for BioMedical Research, Basel, Switzerland.1Department of Biosystems Science and Engineering, ETH Zurich, Basel, Switzerland.Lindner 2† Vanessa Cornacchione 2 Mariavittoria Iazeolla 2 Enkelejda Miho 1 Andreas Zingg 1 Simon Meng 1 Elisabetta Traggiai 2* Sai T.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
